Mildronate® (500 mg) Meldonium
I kēiaʻatikala, hiki iā ʻoe ke heluhelu i nā ʻōkuhi no ka hoʻohana ʻana i ka lāʻau lapaʻau Mililiana. ^ E Ha yM. Hāʻawi i ka hoihoi mai ka poʻe kipa i ka pūnaewele - nā mea kūʻai o kēia lāʻau lapaʻau, me nā manaʻo o nā loea lapaʻau e pili ana i ka hoʻohana ʻana iā Mildronate i kā lākou hoʻomaʻamaʻa. ʻO kahi noi nui, ʻo ia ke hoʻoikaika ikaika i kāu mau loiloi e pili ana i ka lāʻau lapaʻau: kōkua ka lāʻau lapaʻau a ʻaʻole hoʻi i kōkua i ka hoʻopiʻi ʻana i ka maʻi, nā mea hoʻopiʻi a me nā hopena ʻaoʻao, ua ʻike ʻia, ʻaʻole paha e hoʻolaha ʻia e ka mea hana ma ka palapala. Nā kikowaena o Mildronate ma ke alo o nā mea hoʻohālike e pili ana. Hoʻohana e mālama i ka hoʻoulu ʻana o ka puʻuwai a me nā hōʻeha a hoʻomaikaʻi i nā metabolism i loko o nā pā i nā pākeke, nā keiki, me ka wā hāpai a me ka lactation.
Mililiana - kahi lāʻau lapaʻau e hoʻomaikaʻi i ka metabolism. ʻO Meldonium (ʻo ka hana ikaika o ka lāʻau Mildronate) kahi mea hoʻopili hangaluma o ka gamma-butyrobetaine, kahi mea i loaʻa i kēlā me kēia cell o ke kino o ke kanaka.
Ma lalo o nā kūlana o ka hoʻonui ʻana i ka ukana, hoʻihoʻi hou ʻo Mildronate i ke kaulike ma waena o ka lawe ʻana a me ka koi o ka oxygen cells, e hoʻopau ana i ka hōʻemi ʻana o nā huahana mea kūlohelohe i nā cell, pale iā lākou mai ka pōʻino, a loaʻa iā ia kekahi hopena tonic. Ma muli o kona hoʻohana, loaʻa i ke kino ke kū i ka ukana a hoʻihoʻi koke i nā mālama kumu. Ma muli o kēia mau waiwai, hoʻohana ʻia ʻo Mildronate e mālama i nā ʻano like ʻole o ka ʻōnaehana cardiovascular, ka hāʻawi ʻana o ke koko i ka lolo, a me ka hoʻonui ʻana i ka hana kino a me ka noʻonoʻo.
Ma muli o ka emi ʻana o ka hiki ʻana o ke kukū carnitine, ʻoi aku ke ʻano o ka hōʻailona gamma-butyrobetaine me nā vasodilating. Ma ka hopena o ka pōʻino myocardial ischemic, hoʻopaneʻo Mildronate i ka hana ʻana o ke aniani necrotic, e pōkole ana i ka manawa o ka hoʻoponopono.
I ka hana ʻole o ka naʻau, hoʻonui i ka lāʻau i ka myocardial contractility, hoʻonui i ke ahonui i ka hoʻohaʻahaʻa ʻana, a me ka hōʻemi ʻana i ka wā o ka hoʻouka ʻana o ka angina.
Ma ka maʻi ʻoi a me ka maʻi ischemic e pili ana i ka hoʻomaʻamaʻa cerebral e hoʻomaikaʻi i ke kahe o ke koko i ka neʻe ʻana o ischemia, ua hoʻopiʻi i ka hoʻokaʻawale ʻana i ke koko i ka makemake i ka wahi ischemic.
Hoʻohana pono no nā vascular a me dystrophic pathology o ka waihona kālā.
Hoʻomaʻa ʻia ka lāʻau lapaʻau i nā hana hana o ka hoʻopuni o ka hoʻopalapula, hoʻomohala i ke ala o ke komo ʻana i ka nui o ka waiʻona i nā mea maʻi me ka maʻi alakaʻawa me ka wehe ʻokoʻa.
Lapaʻau lāʻau
Ma hope o ka lawelawe waha, ua paʻa koke ka lāʻau mai ka hoʻoili ʻana. Hoʻomili ʻia ia i ke kino me ke hoʻokumu ʻana o ʻelua mau metabolites nui i hoʻopā ʻia e nā keiki.
Nā Hōʻike
- i ka hana paʻakikī o ka maʻi naʻau coronary (angina pectoris, myocardial infarction), ʻeha puʻuwai hemaʻa, palaka cardiomyopathy,
- i ka hopena paʻakikī o nā maʻi a me ka maʻi cerebrovascular maʻi (hōʻeha a me ka cerebrovascular insufficiency),
- hoʻemi ʻia ka hana
- ke kaumaha kino (me ka waena o nā mea hoʻokūkū)
- withdrawal syndrome i loko o ka palaka palaka (me ka hui pū me nā lāʻau kūikawā no ka waiʻona),
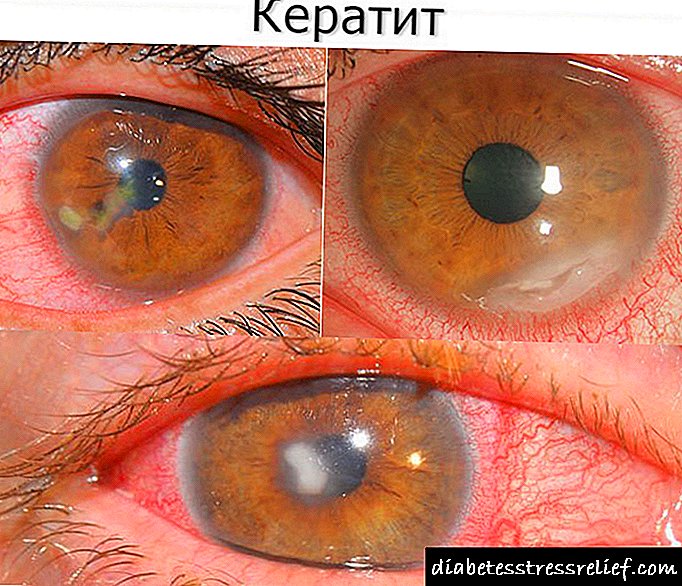
- hemophthalmus, hemorrhages retinal o nā etiologies o nā ʻano,
- he thrombosis o waena o ka veina a i kona mau lala,
- retinopathies o kēlā me nā ʻano etiologies (maʻi maʻi, hypertonic).
E hoʻokuʻu i nā palapala
Nā Capsules 250 mg a me 500 mg (i kekahi manawa i kuhihewa ʻia i nā papa, akā ʻaʻole i noho ka papa papa o Mildronate)
Ke kūmole no ka intravenous, intramuscular a me ka injections parabulbar (injections ma ampoules).
Nā ʻōkuhi no ka hoʻohana a me ka helu ʻana
I ka pilina me ka hiki ke hoʻomohala ʻana i kahi hopena hōʻeuʻeu, ua pono ke inu ʻia i ka lāʻau i ke kakahiaka.
No nā maʻi cardiovascular ma ke ʻano he lāʻau paʻakikī, ua kuhikuhi mua ʻia ka lāʻau lapaʻau ma kahi o ka 0.5-1 g i kēlā me kēia lā, ʻo ka nui o ka hoʻohana ʻana he 1-2. ʻO ke ʻano o ka mālama ʻana he mau wiki 4.
Me ka cardialgia e kū'ē ana i kahi kūlana o ka dystrophy myocardial dishormonal, ua kuhikuhi ʻia ʻo Mildronate ma nā waha he 250 mg 2 mau manawa i ka lā. ʻO ke ala no ka mālama ʻana he 12 mau lā.
I ka hihia o ka ulia cerebrovascular ma ke keaka, e mālama ʻia ka lāʻau lapaʻau ma ka intravenously (ma ke ʻano kūpono no ke ʻano - 500 mg hoʻokahi lā i ka lā 10 mau lā), a laila huli lākou e lawe i ka lāʻau lapaʻau i loko e ka 0.5-1 g i kēlā me kēia lā. ʻO ka papa aoao o ka maʻiʻo 4-6 mau pule.
I loko o nā pilikia o ka hoʻopukapuka o ka cerebral, e lawe ʻia ka lāʻau lapaʻau ma ka waha he 0.5-1 g i kēlā me kēia lā. ʻO ka papa aoao o ka maʻiʻo 4-6 mau pule. ʻ arelelo ʻia nā papa hana e like me ka 2-3 i hoʻokahi makahiki o ka makahiki.
No ka hoʻomaʻamaʻa noʻonoʻo a me ke kino, ua kuhikuhi ʻia i loko o 250 mg 4 mau manawa i ka lā. ʻO ke ala e mālama ai he 10-14 mau lā. Inā kūpono, hoʻomaʻamaʻa ʻia ka lāʻau ma hope o 2-3 mau pule.
Aʻa ʻia nā mea ʻāpono i ka hoʻohana ʻana i ka 0.5-1 g 2 mau manawa i ka lā ma mua o ke aʻo ʻana. ʻO ka lōʻihi o ka papa i ka wā hoʻomākaukau no 14-21 lā, i ka manawa o ka hoʻokūkū - 10-14 lā.
Ma ka palaka hoʻohālikelike, kuhikuhi ʻia ka lāʻau lapaʻau ma ka waha ma 500 mg 4 mau manawa i ka lā. ʻO ke ala e mālama ai he 7-10 lā.
No nā maʻi cardiovascular ma ke ʻano o ka hōʻemi paʻa o ka lāʻau, kuhikuhi ʻia ka lāʻau lapaʻau ma kahi o ka 0.5-1 g i kēlā me kēia lā intravenously (5-10 ml o kahi hoʻonā wai pū me ka neʻe o 500 mg / 5 ml), ʻo ka pinepine o ka hoʻohanaʻana he 1-2 manawa i ka lā. ʻO ka papa hana o ka lapaʻau he 4-6 mau pule.
Ma ka hihia o ka ulia cerebrovascular ma ke ao, ua mālama ʻia ka lāʻau lapaʻau iv 500 mg hoʻokahi i ka lā no 10 mau lā, a laila huli lākou e lawe i ka lāʻau lapaʻau i loko (i loko o ka puka ʻai kūpono, 0.5-1 g i kēlā me kēia lā). ʻO ka papa aoao o ka maʻiʻo 4-6 mau pule.
Ma ka hihia o ka vascular pathology a me dystrophic retinal maʻi, Mildronate e kāhea ana i ka parabulbarly ma kahi o ka olelō he 0.5 ml no ka maʻi ʻaina me ka paona o 500 mg / 5 ml no 10 mau lā.
No ka noʻonoʻoʻana i ka noʻonoʻo a me ke kino, ua kauoha ʻia ka iv mg 500 mg hoʻokahi i ka lā. ʻO ke ala e mālama ai he 10-14 mau lā. Inā kūpono, hoʻomaʻamaʻa ʻia ka lāʻau ma hope o 2-3 mau pule.
Ma ka palaka kūlohelohe, ua kauoha ʻia ka lāʻau lapaʻau iv 500 mg 2 mau manawa i kahi lā. ʻO ke ala e mālama ai he 7-10 lā.
Loaʻa ka hopena
- tachycardia
- nā hoʻololi i ke koko
- psychomotor agitation,
- lie kanikela
- nā hōʻailona dyspeptic
- nā maʻi kūloko (ʻulaʻula o ka ʻili, ke ʻokeo a i ʻole paha, ka ʻeha o ka ʻili, ke kuapo ʻana),
- nāwaliwali laulā
- ka māla ʻana.
Nā Hoʻohui
- hoonui ka maʻi intracranial (komo i nā hihia o ka laha o ka mana o ka anuu, i loko o nā maʻi o ka maʻi intacranial),
- na keiki a me na opio malalo o na makahiki 18
- hypersensitivity i ka lāʻau.
ʻŌpū a me ka lactation
ʻAʻole i ʻike ʻia ka palekana o ka hoʻohana o Mildronate i ka wā hāpai. I mea e pale aku ai i kahi hopena maikaʻi loa i ka wā hāpai, ʻaʻole pono ke kuhikuhi ʻia ka lāʻau lapaʻau i ka wā hāpai.
ʻAʻole i ʻike ʻia a hoʻopā ʻia ʻo ia mea lapaʻau ma ka umauma wai. Inā pono e hoʻohana i ka Mildronate i ka wā lactation, pono ke hōʻuluʻulu i ka ʻōpū.
Nā ʻōlelo kikoʻī
ʻO nā mea maʻi me ka maʻi kūlohelohe a me nā maʻi maʻi e akahele me ka hoʻohana lōʻihi ʻana o ka lāʻau. Inā makemake ʻoe i ka lōʻihi (ma mua o hoʻokahi mahina) e hoʻohana ai i ka lāʻau lapaʻau, pono ʻoe e ʻimi i kahi loea.
I nā makahiki he nui o ke ʻike ʻana i ka mālama ʻana i ka maʻi myocardial infarction a me ka angina hiki ʻole i ka keʻena cardiology hōʻike ʻo Mildronate ʻaʻole ia ka lālani mua no ka maʻi coronary syndrome.
Hoʻohana ʻo Pediatric
I nā keiki a me nā ʻōpio ma lalo o ka makahiki 18, ʻaʻole i hoʻokumu ʻia ka pono a me ka palekana o Mildronate ma ke ʻano o nā capsules a me ka injection.
Hoʻokomo i ka hiki ke lawe i nā kaʻa a kāohi i nā mīkini
ʻAʻohe mea iʻike i ka hopena hewa o Mildronate ma ka helu o ka hopena psychomotor.
Hoʻohui nūhou
Ke hoʻohui, hoʻohui ʻo Mildronate i ka hana o nā lāʻau antianginal, kekahi mau lāʻau antihypertensive, glycosides cardiac.
Hiki ke hui pū me ka Mildronate me nā lāʻau antianginal, anticoagulants a me nā ʻenehana antiplatelet, nā lāʻau antiarrhythmic, diuretics, bronchodilators.
Ke hui pū me ka Mildronate nitroglycerin, nifedipine, alpha-blockers, antihypertensive drug a peripheral vasodilators, tachycardia maʻalahi, hiki i ka arterial hypotension e hoʻomohala (pono e hoʻomaʻamaʻa i ka wā e hoʻohana ai i kēia hui pū).
Nā hōʻike o ka lāʻau Mildronate
Hoʻololi nā kumu hoʻohālike o ka waiwai ikaika:
- 3- (2,2,2-Trimethylhydrazinium) nā mea i kuʻi ʻia dihydrate,
- ʻO Vasomag
- Idrinol
- Kāula
- Medera
- Meldonium
- Meldonius Eskom
- Meldonia dihydrate,
- Kelow,
- Midolat
- Trimethylhydrazinium propionate dihydrate.
Pākuʻi helu
Aia i hoʻokahi kapsul
mea waiwai - meldonium dihydrate 500 mg,
nā mea hoʻohālikelike: ka ʻōpala maloʻo, koloneida polikona, ka calcium stearate,
kapili (kino a me ke kapa): titanium dioxide (E 171), gelatin.
Nā hopuhina gelatin kīnā 00. Keʻokeʻo. ʻO ke kiko kahi paura keʻokeʻo keʻokeʻo me kahi ʻala maʻū. ʻO ka pauka he hygroscopic.
ʻO nā waiwai Pharmacological
Lapaʻau lāʻau
Ma hope o kahi hoʻokahi hoʻokele waha o meldonium, ʻo ka nui o ka plasma wela (Cmax) a me ka mahele o ka pae o ka cur-time curry (AUC) e hoʻonui i ka kaomi i ka ʻoka i kau ʻia. ʻO ka manawa e hiki ai i ka plasma kiʻekiʻe (tmax) ka 1-2 hola. Me ka hana hoʻihoʻi hou ʻia, ʻike ʻia ka hoʻāla ʻana o ka protein equilibrium i loko o nā hola 72-196 ma hope o ka noi ʻana o nā kuʻuna mua. ʻO ka hōʻiliʻili ʻana o ka meldonium i loko o ke kaila koko he hiki. Hoʻololi ka meaʻai i ka loaʻa ʻana o ka meldonium me ka ʻole o ke hoʻololi ʻana iā Cmax a me AUC.
Hoʻonui koke ʻo Meldonium mai ke kahawai e pili ana i nā ʻili. Hoʻonui ka paʻū ʻana i ka protein protein me ka manawa ma hope o ka pau ʻana o ka dosis. ʻO Meldonium a me kona metabolites kahi i lanakila ma ka pale i wahi. ʻAʻohe hana i hōʻike ʻia o ka excretion o meldonium i loko o ka umauma umauma o ke kanaka.
Hoʻololi ka nui o ka meldonium i loko o ke akea.
ʻO Rcr excretion e hana i ka waiwai nui ma ka excretion o meldonium a me nā metabolites. ʻO ka hoʻopauʻana hapa-hapalua o ka meldonium (t1 / 2) ma kahi o 4 hola. Me nā kānana maʻamau, ʻokoʻa ka hapalua o ke ola.
Nā hui ahonui kūikawā
ʻO nā maʻi maʻi
Hiki ke hoʻēmi ʻia ka nui o ka meldonium i nā poʻe maʻi me nā maʻi i ʻole aʻeha o ka naʻau a me ka hana ʻana paha, a ua nui ka nui o ka bioavailability.
ʻO ka maʻi me ka hana renal impaired
ʻO nā mea maʻi me ka hana renal impaired, ka nui o nā bioavailability e ʻike nui ʻia, e hoʻemi i ka ʻōpū. Aia kahi hui o ka renab reabsorption o ka meldonium a i ʻole nā metabolites (no ka laʻana, 3 - hydroxymeldonium) a me ka karnitine, ma muli o ka hoʻomaʻemaʻe kino āpau o ka hoʻonui i ka maʻi karnitine. ʻAʻohe hopena pololei o ka meldonium, GBB, a me ka hui pū ʻana o ka meldonium / GBB ma ka ʻōnaehana renin-angiotensin-aldosterone.
ʻO nā mea maʻi me ka hana pūlima hemahema
ʻO nā mea maʻi me ka pīhoihoi hana ʻino, ka mea i loaʻa i ka bioavailability e ʻike nui, e hoʻemi i ka nui o ka meldonium. ʻO ka hoʻololi ʻana i loko o ka hana akina i loko o nā kānaka ma hope o ka noi ʻana o nā papa he 400-800 mg ʻaʻole i mālama ʻia. Hiki ke hoʻoili ʻia ka hoʻopili ʻia o nā momona i nā pūpū o nā leʻa.
ʻAʻohe ʻikepili e pili ana i ka palekana a me ka hana pono o ka hoʻohana ʻana o ka meldonium i nā keiki a me nā ʻōpio (ma lalo o ke 18 makahiki o ka makahiki), no laila ke hoʻohana ʻia ʻana o ka meldonium ma kēia pūʻulu o nā maʻi.
Lapaʻau lāʻau
ʻO Meldonium kahi mea pili i ka carnitine, kahi kikoʻī ʻĀpana o gamma-butyrobetaine (GBB), kahi i hoʻohuli ai nā molekone i ke kinolika atom.
Ma lalo o nā kūlana o ka hoʻonui i ka ukana, hoʻihoʻi ka meldonium i ke kaulike ma waena o ka lawe ʻana a me ka koi o ka oxygen cells, e hoʻoneʻe i ka hōʻuluʻulu ʻana o nā huahana mea olakino i loko o nā kōpale, pale iā lākou mai ka pōʻino, a loaʻa iā ia kekahi hopena tonic. Ma muli o kona hoʻohana, loaʻa i ke kino ke kū i ka ukana a hoʻihoʻi koke i nā mālama kumu. Ma muli o kēia mau waiwai, hoʻohana ka meldonium i ka mālama ʻana i nā ʻano like ʻole o ka ʻōnaehana cardiovascular, ka hāʻawi ʻana o ke koko i ka lolo, a me ka hoʻonui ʻana i ka hana kino a me ka noʻonoʻo. Ma muli o kahi o ka emi ʻana o ke kukū carnitine, ʻo GBB, ka mea i loaʻa nā vasodilating, ua hoʻohālikelike ʻia. I ka hihia o ka poʻomaka ischemic poino i ka myocardium, lohi nā meldonium i ka hoʻokumu ʻana o ke rohe necrotic a pōkole i ka manawa hana hoʻoponopono. Me ka puʻuwai puʻuwai, e hoʻonui ai i ka aelike myocardial, hoʻonui i ke ahonui i ka hoʻohaʻahaʻa ʻana, a me ka hōʻemi ʻana i ka ʻike o ka hoʻouka ʻana o angina. Ma ka maʻi ʻoi a me ka maʻi ischemic e pili ana i ka hoʻomaʻamaʻa cerebral e hoʻomaikaʻi i ke kahe o ke koko i ka neʻe ʻana o ischemia, ua hoʻopiʻi i ka hoʻokaʻawale ʻana i ke koko i ka makemake i ka wahi ischemic. Ma ka hihia o nā pilikia neurological (ma hope o nā ulia cerebrovascular, ka hoʻohana ʻana i ka manawa, ka hōʻeha poʻo, ka encephalitis hōʻailona-hopu) e pili pono ana i ke kaʻina hana o ke kino a me nā hana naʻauao i ka wā o ka hoʻihoʻi.
Nā hōʻailona no ka hoʻohana ʻana
I ka hoʻonohonoho paʻakikī i nā pilikia penei:
nā maʻi o ka puʻuwai a me ka ʻōnaehana vascular: ka kaina paʻa, ka paʻa ʻole o ka naʻau (I-III hana papa NYHA), cardiomyopathy, nā hana hoʻokele o ka puʻuwai a me nā ʻōnaehana vascular,
ka huʻi a me ka maʻi ischemic i hoʻopuka ʻia o ka cerebral circulation,
ka hoʻemi ʻana i ka hana, hoʻonaninani kino a me ka psycho-emotional overstrain,
i ka wā e hoʻihoʻi ana mai ka maʻi cerebrovascular, hōʻeha ke poʻo a me ka encephalitis.
ʻLoe a me ke kākele
Pākuʻi i loko. Kuke ʻia ka pahu i ka wai. Hiki ke hoʻohana ʻia ka lāʻau lapaʻau ma mua a ma hope o ka ʻaina. I ka pilina me ka hiki ke kūpono i ka hopena hoʻoikaika ʻia, hoʻolako ʻia ka lāʻau lapaʻau e hoʻohana i ke kakahiaka.
Nā pākeke
ʻO nā maʻi o ka puʻuwai a me nā ʻōnaehana vascular,nā maʻi hake
ʻO ka dosis kahi 500-1000 mg i kēlā me kēia lā. Hiki i ka ʻonaʻohana i kēlā me kēia lā i hoʻokahi manawa a hoʻokaʻawale paha i ʻelua pūmole hoʻokahi. ʻO ka palena kiʻekiʻe loa o kēlā me kēia lā he 1000 mg.
Hoʻemi ka hana, overstrain a me ka wā hoʻolaʻa
ʻO ka dosis kahi 500 mg i kēlā me kēia lā. ʻO ka palena kiʻekiʻe loa o kēlā me kēia lā he 500 mg.
ʻO ka lōʻihi loa o ka mālamaʻana ʻo 4-6 mau pule. Hiki i ke ʻano o ka mālama ʻana i nā manawa 2-3 i ka makahiki.
ʻO nā maʻi maʻi
ʻO nā mea maʻi aʻokiʻoki me ka pōpoki a me / a i ʻole ka hana ʻana o ka pūpū e pono ai e hōʻemi i ka nui o ka meldonium.
ʻO ka maʻi me ka hana renal impaired
No ka mea ua hoʻōla ʻia ka lāʻau lapaʻau mai ke kino ma o nā keiki, nā poʻe maʻi me ka hana ʻoi aku ka maʻi papalapa mai ka hōʻemi a i ke kaumaha ʻole ʻia e hoʻohana i kahi haʻahaʻa o meldonium.
ʻO nā mea maʻi me ka hana pūlima hemahema
ʻO nā mea maʻi me ka maʻi maʻi o ka maʻi hepatic i mea e hoʻohana ai i kahi haʻahaʻa o ka meldonium.
Ka maʻi ʻōpiopio
ʻAʻohe ʻikepili e pili ana i ka palekana a me ka hana pono o ka hoʻohana ʻana i ka meldonium i nā keiki a me nā ʻōpio i lalo o nā makahiki he 18, no laila ke hoʻohana ʻia ʻana o kēia lāʻau lapaʻau nā keiki a me nā ʻōpio.
Nā hopena hopena
- hypersensitivity, alahula dermatitis, rashes (ka maʻamau / macular / papular), nāwaliwali, urticaria, angioedema, anaphylactic hopena
- Arousal, kahi hopohopo o ka makaʻuʻole, nā manaʻo nānāʻole, ke hiamoe
- paresthesia, hypesthesia, tinnitus, vertigo, dizziness, gait disturbance, nāwaliwali, ka nalowale o ka ʻike
- hoʻololi i ka puʻuwai naʻau, palpitations, tachycardia / sinus tachycardia, atrial fibrillation, arrhythmia, ʻehaʻeha / umauma ʻeha
- ka huki i ka hopena o ke koko, ke kīpē hypertensive, ka maʻiemia, ka paila o ka ʻili
- ʻeha o ka ʻōpū, ʻōpala, dyspnea, apnea
- dysgeusia (ʻoliʻoli ʻōlelo i loko o ka waha), ka nalo ʻana o ka ʻono, ka luaʻi, ka luaʻi, ka luaʻi, ka hōʻuluʻulu ʻana o ka ʻōpū, ʻeha, ka ʻeha o ka ʻōpū, ke ʻeha hope, nāwaliwali o ka naʻau, nā ʻōpū o ka naʻau,
- nāwaliwali laulā, haʻalulu, asthenia, edema, ke kuamoʻo o ka maka, ka ʻōpū o nā wāwae, ka wela ʻana o ka wela, ʻāwīwī o ke anu, ke anu anuanu
- lihilahi i ka electrocardiogram (ECG), ka wikiwiki o ka naʻau, eosinophilia
Nā Hoʻohui
- Nā maʻi maʻi o ka mea ikaika a i ʻole nā mea kōkua o ka lāʻau lapaʻau.
- ke hoʻonui i loko o ke kaomi intacranial (i ka hana hewa ʻole o nā kaila o venous, intracranial tumors).
- ka maʻi hepatic a me / a ʻole ka hana ʻole ʻana i ka pani ʻole ma muli o ka lawa ʻole o ka ʻike palekana.
- ka wahine a me ka lactation, ma muli o ka nele o nā ʻikepili i ka hoʻohana ʻana i nā lāʻau lapaʻau i ka wā o kēia manawa.
- nā keiki a me nā ʻōpio ma lalo o 18 mau makahiki o ka makahiki, ma muli o ka hapa ʻole o nā data i ka hoʻohana ʻana i ka lāʻau lapaʻau i ka wā o kēia manawa.
Hoʻopili i nā lāʻau lapaʻau
Hoʻonui i ka hopena o nā mea hoʻomake coronary, kekahi mau lāʻau antihypertensive, glycosides cardiac.
Hiki ke hoʻohui pū me nā mea antianginal drug, anticoagulants, antiplatelet agents, antiarrhythmic drug, diuretics, bronchodilators.
Hiki i ka meldonium ke hoʻonui i ka hopena o nā lāʻau lapaʻau e loaʻa ana i ka glyceryl trinitrate, nifedipine, beta-blockers, nā lāʻau antihypertensive, a me nā vipodileta peripheral.
I ka poʻe maʻi me ka puʻuwai naʻau ʻole e lawe i ka meldonium a me ka lisinopril i ka manawa like, ua hōʻike ʻia ka hopena maikaʻi o ka hui hoʻohui ʻana (vasodilation o nā kīʻaha nui, ka hoʻomaikaʻi ʻana i ka sirkila peripheral a me ka maikaʻi o ke ola, ka hōʻemi o ka noʻonoʻo a me ke kaumaha kino).
I ka hoʻohana ʻana i ka meldonium i ka hui pū me ka hoʻonaninika orotic e hoʻopau i ka pōʻino i hōʻemi ʻia e ischemia / reperfusion, ua nānā ʻia kahi hopena pharmacological hou.
ʻO kahi hopena o ka hoʻohana like ʻana Sorbifer a me ka meldonium i nā maʻi me ka iron anemia deficiency, ʻo ka hana ʻana o nā mea momona momona i loko o nā kōpela koko ʻokoʻa.
Kōkua ʻo Meldonium i ka hoʻohemo ʻana i nā loli pathological i loko o ka puʻuwai i hoʻokumu ʻia e ka azidothymidine (AZT), a me ka hoʻihoʻi ʻole ʻia i nā hopena kaumaha o ka oxidative i hoʻomake ʻia e AZT, e alakaʻi ana i ka mitochondrial dysfunction. ʻO ka hoʻohanaʻana o ka meldonium i ka hui pū me AZT a i nā mea lāʻau'ē aʻe no ka mālamaʻana i ka immunodeficiency syndrome (AIDS) e loaʻa i kahi hopena kūpono i ka lāʻau maʻi AIDS.
Ma ka hoʻāʻo e hoʻoneʻeneʻe i ka hoʻoneʻe étananololo, hoʻemi ʻo meldonium i ka lōʻihi o ka hiamoe. I ka wā hoʻopiʻi ʻia e ka pentylenetetrazole, ua hoʻokū ʻia kahi hopena anticonvulsant o ka meldonium. I ka huli ana, i ka mea e ʻoi aku ana iā α-adrenoblocker yohimbine ma ke kūkaʻi o 2 mg / kg a me N- (G) -nitro-L-arginine synthase inhibitor ma ke ʻano o 10 mg / kg e hoʻohana ʻia ma mua o ka mālama ʻana me meldonium, ua pau ʻole ka hopena anticonvulsant o ka meldonium. . ^ E Ha yM.
Hiki i kahi overdose o ka meldonium ke hoʻonui i ka cardiotoxicity i kumu e ka cyclophosphamide.
ʻO kahi hemahema carnitine i hopena mai ka hoʻohana ʻana o D-carnitine (ʻo kahi isomer a pharmacologically inactive) -meldonium hiki ke hoʻoikaika i ka cardiotoxicity i kumu o ka ifosfamide.
ʻO Meldonium kahi hopena palekana i ka hihia o ka cardiotoxicity i kumu e ka indinavir a me nā neurotoxicity i kumu e ka efavirenz.
Ma muli o ka hiki ke hoʻomohala ʻana i ka tachycardia maʻalahi a me ka hypotension arterial, pono e hoʻomaʻemaʻe ʻia ka mālama ʻana i ka wā e hui pū ai nā lāʻau lapaʻau e loaʻa ka hopena, me nā ʻano lāʻau ʻē aʻe e pili ana i ka meldonium.
Nā ʻōlelo kikoʻī
Me ka lōʻihi o ka hoʻohana ʻana i ka lāʻau, pono ka poʻe maʻi me ka ʻehaʻeha a me nā maʻi ʻē aʻe e pono ai e makaʻala (ʻū a me / a i ʻole e nānā pono i ka nānā ʻana o ka hana o ka maʻi).
ʻAʻole ʻo Meldonium kahi lāʻau mua-laina no ka maʻi coronary syndrome.
Nā hiʻohiʻona o ka hopena o ka lāʻau lapaʻau ma ka hiki ke hoʻokau i kahi kaʻa a lawelawe paha
Pono e mālama pono i ka wā e lawe ana i kahi kaʻa a i ʻole nā pilikia ʻeha weliweli.
Ke keu
ʻAʻole ʻike maopopo ʻia nā hihia o ka overdose me meldonium, he laʻa ka lāʻau lapaʻau.
Me ke kahe koko haʻahaʻa, pilikia nui, hōʻeha, tachycardia, a me ka nāwaliwali ākea hiki.
I ka hihia o ka overdose koʻikoʻi, pono ia e kāohi i ka hana o ka nui o nā a me nā aʻa.
Ma muli o ka hoʻopili ʻia ʻana o ka lāʻau lapaʻau i nā protein, ʻaʻole nui ka hemodialysis.
Mea hana
ʻO JSC "Nā Mīkini", Latvia
Wili o ka hui e hoʻokipa ana i ka'āinaKe kupa nei ʻo Republic of Kazakhstan mai nā mea kūʻai aku e pili ana i ka maikaʻi o ka huahana
Koho o JSC "Kahiki"
050010, Almaty, Dostyk Ave., kihi o ul. Bogenbai Batyr, d. 34a / 87a, oihana No. 1
Lapaʻau lāʻau
ʻO Meldonium (MILDRONAT ®) kahi mea hoʻohui o ke ʻano gamma-butyrobetaine - kahi waiwai i loaʻa i kēlā me kēia pili o ke kino o ke kanaka.
Ma lalo o nā kūlana o ka hoʻonui ʻana i ka ukana, hoʻihoʻi ka MILDRONAT ® i ke kaulike ma waena o ka lawe ʻana a me ka koi o ka oxygen cells o nā huina, ke kāpae nei i ka hōʻemi ʻana o nā huahana mea olakino i loko o nā pā, mālama iā lākou mai ka pōʻino, a loaʻa iā ia kekahi hopena tonic. Ma muli o kona hoʻohana, loaʻa i ke kino ke kū i ka ukana a hoʻihoʻi koke i nā mālama kumu.
Ma muli o kēia mau waiwai, hoʻohana ʻia ka lāʻau MILDRONAT ® e mālama i nā ʻano like ʻole o ka CVS, ka hāʻawi ʻana o ke koko i ka lolo, a me ka hoʻonui ʻana i ka hana kino a me ka noʻonoʻo. Ma muli o ka emi ʻana o ka hiki ʻana o ke kukū carnitine, ʻoi aku ke ʻano o ka hōʻailona gamma-butyrobetaine me nā vasodilating. I ka hihia o ka hōʻemi kino o ka ischemic i ka myocardium, hoʻopau ʻia ka lāʻau MILDRONAT ® i ka hoʻokumu ʻana i ke kīkoʻo necrotic a pōkole i ka wā hoʻoponopono. Me ka puʻuwai puʻuwai, e hoʻonui ai i ka aelike myocardial, hoʻonui i ke ahonui i ka hoʻohaʻahaʻa ʻana, a me ka hōʻemi ʻana i ka ʻike o ka hoʻouka ʻana o angina. Ma ka maʻi ʻoi a me ka maʻi ischemic disorders o ka cerebral sirkulation, ʻo ka lāʻau lapaʻau MILDRONAT ® e hoʻomaikaʻi i ke kahe koko i loko o ka manaʻo o ischemia, paipai i ka hoʻokaʻawale ʻana o ke koko i ka makemake i ka wahi ischemic. Hoʻomaʻa ʻia ka lāʻau lapaʻau i nā hana hana o ka palupene i loko o nā mea maʻi me ka maʻi kūlohelohe me ka maʻi withdrawal.
Nā hōʻikeʻike o ka lāʻau MILDRONAT ®
ulia ʻana o ka maʻi coronary maʻi maʻi (angina pectoris, myocardial infarction),
ka huhū puʻuwai a me ka cardiomyopathy ma ke kua ʻana o ka maʻi hormonal,
uluhua o ke oola a me nā pilikia ke kakaki o ka hoʻokahe koko i ka lolo (stroke and cerebrovascular insufficiency),
ʻophpopa hemophthalmus a me retinal heminal o nā ʻano etiologies, thrombosis o ke ake retinal vein a me kāna mau lālā, retinopathy o nā ʻano etiology (maʻi, maʻi maʻi, hypertension),
ka manaʻo noʻonoʻo a me ke kino (me ka poʻe hana ʻoihana) (hiki i ka lāʻau lapaʻau ke hāʻawi i kahi hopena kūpono i ka wā e lawe ana i ka control doping (ʻike. "ʻōlelo aʻoi aku")
withdrawal syndrome i loko o ka palaka palaka (i hui pū me ka lāʻau kūikawā no ka waiʻona).
ʻŌpū a me ka lactation
ʻAʻole i aʻo ʻia ka palekana o ka hoʻohana ʻana i nā wahine hāpai, no laila ke contraindicated hoʻohana ʻia e pale aku i nā hopena pōpilikia i ka maʻi wahine.
ʻO ka hōʻike o ka lāʻau MILDRONAT ® me ka waiū a me kona hopena ma ke kūlana olakino o ka mea hou i aʻo ʻole ʻia, no laila, inā pono, pono e oki ʻia ka mālama ʻana o ka umauma.
Hoʻohui
Hiki ke hoʻohui pū me nā mea hana antianginal, anticoagulants, nā ʻenehana antiplatelet, nā lāʻau antiarrhythmic, diuretics, bronchodilators.
Hoʻonui i ka hana a nā glycosides cardiac.
Ma muli o ka hiki ke hoʻomohala ʻana i ka tachycardia maʻalahi a me ka hypotension arterial, pono e hoʻohana ʻia ka akahele i ka wā e hui pū me ka nitroglycerin, nifedipine, alpha-blockers, nā mea kanu antihypertensive a me nā vasodilators peripheral, Hoʻonui ka MILDRONAT ® i kā lākou hopena.
E hoʻokuʻu i ke ʻano
ʻO ka hoʻonā no ka olakoli intramuscular, intravenous a parabulbar, 100 mg / ml. 5 ml i kahi pahu kīʻaha o ka ʻaoʻao o ka lau o ka papa hydrolytic I me kahi laina a hiki paha kahi hiwahiwa.
5 pākahi pākahi. i loko o nā paʻi polū i hana ʻia me ka film PVC a i ʻole uncoated PET film (pallet). Ma ka 2 a i ʻole (4 no nā mea hana o ZAO Santonika a me ka HSBC Pharma sro) mau ʻāpana cell (pallets) i kahi kāwele pepa.